Why Use Hormone Pellets And Creams
When hormones are absorbed rather than swallowed, they go directly to the tissues and are not altered by the liver. This is called the first pass effect. Bypassing the gastrointestinal system also avoids many possible side effects. We use pellets as a convenient and measurable way to provide your hormone replacement.
Is Bioidentical Hormone Replacement Therapy Approved By The Fda
This therapy is a not a new approach to hormone replacement it has been used since the 1930s. Bioidentical hormones are legal to prescribe and use in the US, although the FDA has not given its specific approval. The lack of FDA approval is because there are no official placebo-controlled studies to prove whether bioidentical hormones are safer than standard hormone replacement therapy.
What Is The Procedure
A healthcare professional will consider factors such as hormone levels and body mass index when working out an appropriate dose of hormones to insert.
To insert hormone pellets, a healthcare professional will numb the skin and make a small incision, usually into the hip area. They will then insert one or more hormone pellets underneath the skin.
They will close the incision with Steri-Strips, a stitch, or both. A person will need to follow the healthcare professionalâs instructions after the procedure. Those with Steri-Strips may be able to remove them after 5â7 days.
The pellets provide a constant supply of hormones for about 3 months.
People wishing to use compounded bioidentical hormones may need to undergo a salivary test first to check their hormone levels. However, according to The American College of Obstetricians and Gynecologists , âsalivary testing does not currently offer an accurate or precise method of hormone testing.â
There is little scientific evidence to support the benefits of hormone pellet therapy. The FDA does not regulate compounded pellet therapy and has not approved it.
Providers of hormone pellet therapy suggest that the treatment will provide various benefits. These include:
- protecting against health conditions, such as heart disease and osteoporosis
There may also be advantages of a pellet delivery system, including:
The ACOG recommends FDA-approved hormone therapy, which includes:
Read Also: How To Boost Estrogen Naturally
Do Bioidentical Hormones Cause Weight Gain
Yes, weight gain is a side effect of bioidentical hormones.
A note from Cleveland Clinic
Bioidentical hormones are used to help people manage symptoms of menopause or other hormonal imbalances. The FDA does not approve some bioidentical hormones. All hormone replacement therapy comes with risks. Compounded bioidentical hormones may come with more risk because their effects are not well studied. Talk to your healthcare provider about the pros and cons of hormone replacement. Most importantly, know that you do not need to live with unpleasant symptoms of low hormones. Your provider can help safely manage your symptoms.
Last reviewed by a Cleveland Clinic medical professional on 04/15/2022.
References
Symptoms And Effects Of Menopause
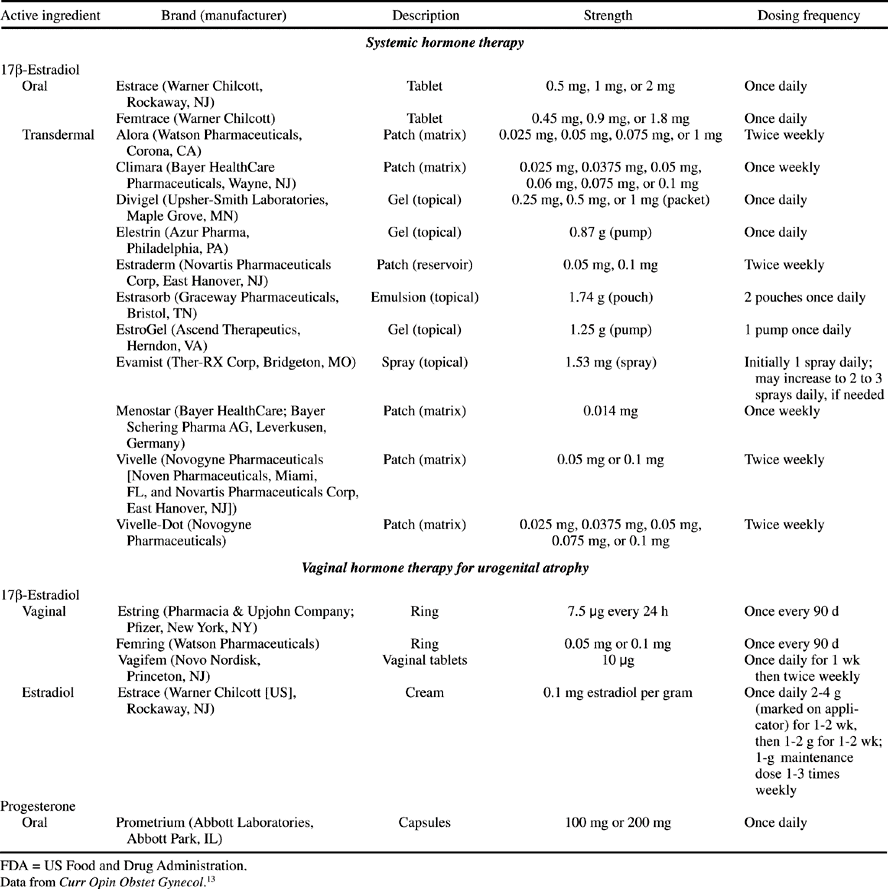
Menopause is the time in a womans life when menstruation ceases, signaling the end of her reproductive ability. The timing of menopause varies widely, but this event often occurs naturally in women in the fourth or fifth decades of life, at a mean age of 51 years. Certain medical or surgical conditions may induce the cessation of menses before this age. If menopause occurs before the age of 40 years, it is considered premature.
The STRAW classification proposed by the American Society for Reproductive Medicine depicts the natural transition in a females life from the reproductive years to the time of menopause.
The reproductive years are divided into early, peak, and late and are characterized by regular menstrual cycles . This is followed by the stage of menopausal transition, which earlier on is characterized by a variable cycle length that is more than 7 days different from normal. During the latter stages of this transition phase, women experience intervals of amenorrhea of more than 60 days. When this duration of amenorrhea lasts for up to 12 months, it is classified as postmenopause. The stage of perimenopause spans from the beginning of the stage of menopause transition up until the completion of 1 year following the final menstrual period.
Don’t Miss: How To Fix Hormonal Acne Naturally
Are Bioidentical Hormones Safe
The bioidentical hormones that are approved by the FDA have been tested for safety. They have passed the FDA’s strict standards and are safe for people to use. Like all hormone treatments, there are risks involved. You should weigh the pros and cons of even the FDA-approved bioidentical hormones with your healthcare provider.
Cme Questions About Bioidentical Hormone Therapy
Don’t Miss: What Are Human Growth Hormones
Claim : Custom Cbht Contains Safer Ingredients Than Fda
The production of US Pharmacopeia ingredients begins with the extraction of diosgenin from plants such as soy and yams.32 A chemical conversion is then required to produce progesterone, which is the precursor to the estrogens and the androgens that are used to formulate the final compounded product.32 This derivation and production process is similar to that of many commercially available, FDA-approved BHT products for which safety and efficacy data are available.
Donna Locke: Fda Wants To Restrict Compounded Bioidentical Hormones
Get ready for more government overreach unless the public blocks the Food and Drug Administrations years-long attempt to mostly prohibit compounding pharmacies from making individual, customized bioidentical hormone products.
Who would benefit from this elimination of competition in the hormone-therapy field? Why, that would be the big pharmaceutical manufacturers who make their own bioidentical hormone products. Yes. Big Pharma, which has put so many of us through body-crashing hell. Ill get to that in a minute.
The FDA says the customized compounded products are a public health concern, and points to FDA-commissioned studies that compounding pharmacists and their advocates say are flawed, biased, incomplete studies done by researchers unfamiliar with compounded hormones or even compounding. Some members of the most recent FDA study and review commission were recommended by the FDA and had close ties to large drug manufacturers opposed to compounded hormones, according to the Alliance for Pharmacy Compounding, a member and advocate group, and other sources.
The FDA says the study found minimal oversight of compounded bioidentical hormones, and a lack of good clinical evidence for their usefulness. My friend who ran out of her compounded hormones and had to stick her head in her freezer to relieve the ensuing hot flashes thinks theyre useful.
You May Like: How To Get Rid Of Adult Hormonal Acne
Why Aren’t Compounded Bioidentical Hormones Fda Approved
The FDA doesn’t approve any compounded products, for any condition, because those products aren’t standardized.
That doesn’t mean that compounding is bad. Compounding can be useful for patients who are allergic to an additive in an FDA-approved product, says Kathleen Uhl, MD, the FDA’s assistant commissioner for women’s health.
But “the purpose of compounding is to do it on a patient-by-patient basis, so there’s nothing that’s submitted to FDA to evaluate, so they’re not FDA approved,” Uhl explains.
And because compounded products don’t go through the FDA approval process, they don’t bear the same warnings as other hormone therapy.
A woman who gets a prescription for an FDA-approved hormone therapy for menopausal symptoms is “going to get a lot of warning information,” but if she gets a compounded product instead, “you don’t get any of those warnings,” Uhl says. “There’s no requirement for them to provide that because those products are not FDA approved.”
L.D. King, executive director of the International Academy of Compounding Pharmacists, suggests that patients look for accredited compounding pharmacies listed on the web site of the Pharmaceutical Compounding Accreditation Board .
Manson points out that with FDA-approved “bioidentical” drugs available, “most women interested in bioidentical formulations do not need to take custom-compounded products .”
Claim : Compounding Provides Improved Delivery And Tolerability Of Ht
Compounded medications require a written prescription from a licensed physician. Prescriptions filled in a compounding pharmacy are prepared, mixed, and assembled according to the specifications of the prescriber. Compounding plays an important role in providing drugs to meet the individualized needs of patients that cannot be met by a commercially available FDA-approved preparation. It may provide a different strength, dose, or delivery system, or it may allow a medication that is otherwise poorly tolerated to be essentially reformulated and delivered in an alternative form .28,29 Compounded hormone preparations are not required to undergo the rigorous safety and efficacy studies required of FDA-approved HT and can demonstrate wide variation in active and inactive ingredients.5
The oversight of accredited compounding pharmacies falls to state pharmacy boards, which are to direct the method of preparation and enforce standards for compounding as dictated by the US Pharmacopeia. Thus, most compounded products do not have scientifically rigorous clinical testing for either safety or efficacy.5,30 A limited FDA survey in 2001 analyzed 29 product samples from 12 compounding pharmacies. None of the products failed identity testing however, 10 of the 29 failed 1 or more quality test, and 9 of the 10 failing products also failed potency or assay testing. For FDA-approved therapies, the usual comparison failure rate is less than 2%.28
Read Also: Do Hormone Pellets Help You Lose Weight
Are Bioidentical Hormones Fda Approved
by DonnaWhite | Oct 11, 2019
The quick answer is yes, bioidentical hormones are FDA approved but hold on, this is a very controversial and somewhat complex topic that requires more explanation. Some bioidentical hormones are manufactured by drug companies, are approved by the FDA, and sold in standard doses. On the other hand some bioidentical hormone preparations are made at special pharmacies called compounding pharmacies. These compounded hormones are made on a case-by-case basis for each patient as ordered by licensed prescribers. Since compounded bioidentical hormones are custom-made for each individual patient, they arent approved by the FDA nor are they required to be. However, they do contain FDA approved hormones in the formula.
Traditional Hrt Vs Bhrt

The goal of hormone replacement therapy is to replace low levels of hormones. While levels may not be completely restored, treatment is usually enough to resolve symptoms. Differences between HRT and BHRT are:
- Traditional HRT refers to synthetic hormones made in the lab. They are FDA-approved medications that come in standard, prepackaged doses.
- BHRT uses synthetic hormones that are the most identical to those in the body. They come in standard, commonly used doses that are approved by the FDA. The chemical structure of the hormones used in BHRT is often the same or similar as those used in traditional therapy.
Read Also: Go With The Flow Hormone Balance For Women
Even Celebrities Believe In Hormone Therapy
Her natural and rich hair may be at least partially based on the bioidentical hormone therapy she has been receiving, something which, according to the article, is also being used by famous names. Billionaire philanthropist Oprah Winfrey, legendary entertainer Madonna, and Oscar-winning actress Angelina Jolie have all used them for reasons such as dealing with the effects of menopause.
Bioidentical hormone replacement therapy to regulate the normal fluctuations of the body may be like a fountain of youth of sorts, maintaining the body and mind at healthy levels, while preventing the aging process by extending the natural development of collagen, keratin and elastin in the skin. These are essential proteins that all play roles in keeping the skin looking youthful.
To understand this a little better, lets briefly examine a womans biology: four major sex hormones are released by the female body: estradiol peaks in the first half of the menstrual cycle of a woman which may lead to a spike in progesterone following ovulation. Testosterone is produced by the adrenal glands and ovaries. Finally, the most abundant sex hormone released during younger years, dehydroepiandrosterone is tied to energy levels as well as total health. Levels of these hormones all waver throughout life as a person ages.
What Are Bioidentical Hormones
Bioidentical hormones are processed hormones designed to mimic the hormones made by your bodys glands. Taking bioidentical hormones can help people who experience symptoms of low or unbalanced hormones. This is often the case for people experiencing symptoms of perimenopause or menopause.
Hormones are chemicals made by your endocrine glands. They are messengers that tell other parts of your body how and when to work. Hormones affect many systems and functions in your body. Even the slightest imbalance can cause symptoms that interfere with your day. Healthcare providers may recommend hormone replacement therapy as a treatment for these symptoms.
Bioidentical hormone therapy uses processed hormones that come from plants. Estrogen, progesterone and testosterone are the most commonly used bioidentical hormones.
Some prescription forms of bioidentical hormones are premade by drug companies. The U.S. Food and Drug Administration has approved certain types of bioidentical hormones. Other forms of bioidentical hormones are custom-made by a pharmacist based on a healthcare provider’s prescription. These are compounded bioidentical hormones.
The compounded forms have not been tested and approved by the FDA. Though it is often advertised that products that are made from plants are “natural” choices, they are altered in a lab so are no longer natural when done with processing.
Conventional hormone therapy vs. bioidentical hormone therapy
Also Check: What Are Some Of The Side Effects Of Melatonin
Neither Natural Nor Safe: Compounded Bioidentical Hormones Need Better Evidence
A category of hormone treatments often promoted as safe and “natural” need far more study and research to evaluate those claims, according to a UConn expert.
Claims about a popular category of hormone treatments are misleading or based on anecdotal evidence rather than rigorous clinical testing.
Women in menopause, men with low testosterone, and adults transitioning genders all sometimes see doctors about hormone therapy. But personally tailored hormone treatments may not be better, or safer, than standard Food and Drug Administration-approved hormone therapies, the National Academies of Science, Engineering and Medicine reported on July 2. Custom hormone treatments, called compounded bioidentical hormone therapies , are often promoted by celebrities as natural or safer than FDA-approved therapies. UConn toxicologist José Manautou, who heads the department of pharmaceutical sciences, served on the committee that wrote the National Academies report. Below, he answers UConn Todays questions about cBHTs and explains why both doctors and consumers should think twice about prescribing and using them.
What does bioidentical mean, and what is a bioidentical hormone therapy?
What conditions are treated with bioidentical hormone therapies?
Primarily symptoms associated with menopause in women. Night sweats, hot flashes and flushes, vaginal atrophy and dryness. Gonadal dysfunction in men is another indication for the use of bioidentical hormones.
Claim : Compounded Bht Is Safer Than Fda
Proponents of CBHT claim that compounded products containing plant-derived hormones that are bioidentical to those found in the human ovary offer better safety, efficacy, and tolerability than noncompounded, FDA-approved HT.23 Unfortunately, these claims have led to confusion among patients, who often look to their physicians for direction. Therefore, physicians who care for women seeking relief from menopausal symptoms need to be familiar with the claims made about custom CBHT products and the evidence in the medical literature on their effectiveness or the lack thereof.
Objective evidence and scientific studies to substantiate these claims are lacking, and the use of custom CBHT vs FDA-approved HT remains a topic of ongoing debate in the lay press, in alternative medicine literature, and in the popular media.15 The claim that the safety profile of bioidentical compounds is better than that of FDA-approved HT15 belies the complexities of the topic. Both CBHT and FDA-approved HT are available in various dosages, combinations, preparations, and routes of delivery that may have differing effects on risk to an individual patient.26
You May Like: How To Have Your Hormones Checked
What We Learned From The Whi
The findings of the E+P arm of the WHI that were published in 2002 dramatically changed the prescribing practices of physicians in the United States.6 Before the trial demonstrated adverse cardiovascular disease events and a 26% increased risk of breast cancer in female participants, the number of women for whom E+P was prescribed had been steadily increasing, from 58 million in 1995 to 90 million in 1999.6 From 1999 to 2002, the numbers stabilized however, within 3 months of publication of the WHI findings, prescriptions for E+P decreased by 63%. Many women stopped HT and some sought out alternative therapies for treatment of symptoms associated with menopause.6 In addition to their effect on physician-prescribing patterns, the WHI findings changed the perceptions of patients about the trustworthiness of the advice dispensed by physicians. A small survey of 97 women conducted in 2005 demonstrated that all the participants had heard of the WHI a substantial number expressed a loss of trust in the information about HT that they received from their physicians.7
What Do Hormone Pellets For Menopause Cost
The cost of hormone pellet therapy can vary depending on the provider, but an average cost may be close to $1,536 each year.
Healthcare insurance may cover FDA-approved hormone therapy, but the extent of the coverage may vary among plans. Most insurance companies are unlikely to cover compounded bioidentical hormone therapy because it is not FDA-approved.
You May Like: Intermittent Fasting For Hormone Belly
Also Check: Does Birth Control Help With Hormones